
Safety Pharmacology Society is a nonprofit organization that promotes knowledge, development, application, and training in Safety Pharmacology—a distinct scientific discipline that integrates the best practices of pharmacology, physiology and toxicology. The 2018 SPS Scientific Program features a diverse range of scientific sessions organized into two themed tracks and covering issues such as
Opiates*, Secondary Pharmacology, Non-opiate Pain Management, Safety Pharmacology/FDA Discussion on GI, CiPA Real-life Implementation, Cardio GPCR*, Cardio-oncology, New Strategies in CNS and Sensory Assessments, Imaging, In Silico, Phase I Trial Design and Translation to Clinical Data, Preclinical to Clinical- Case Studies and much more! This will ensure attendees can stay abreast of new content and developments in key areas of interest.
Stop by
booth #101 to learn how DSI's solutions can improve the data you get from your Safety Pharmacology studies.
Also, visit our neighboring booth #103 to see solutions from Multichannel Systems, a part of our Harvard Bioscience Family.
 Who's Attending?
Who's Attending?



Mariko Austin Mark Kesselem Shannon Leishman
Marketing Manager Senior Account Manager Sales Manager US



Eric Rieux Dan Singer Hiro Takata
VP Global Sales Marketing Director Account Manager
and Marketing
Why DSI?
- Fully implantable telemetry
- Over 30 years of experience in the preclinical research market
- Acquire data from conscious, freely moving laboratory animals
- Stress-free data collection while reducing risk of infection
- Various sizes to support a range of research models (from mice to non-human primates)
- In-house quality and development expertise
- Knowledgeable and responsive global support team that will ensure you have full confidence with your system and acquiring the data you need for your studies.
DSI Products
Cardiovascular Solutions
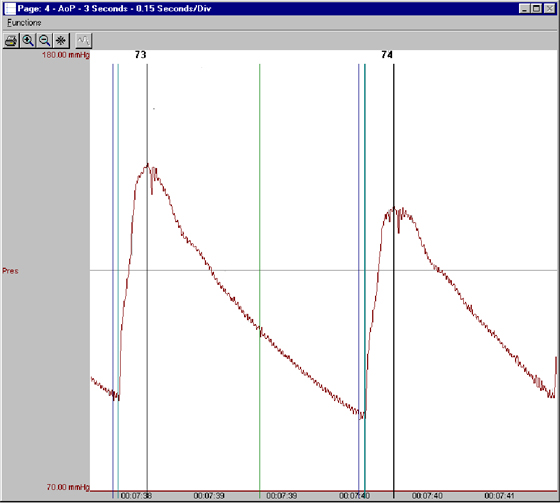
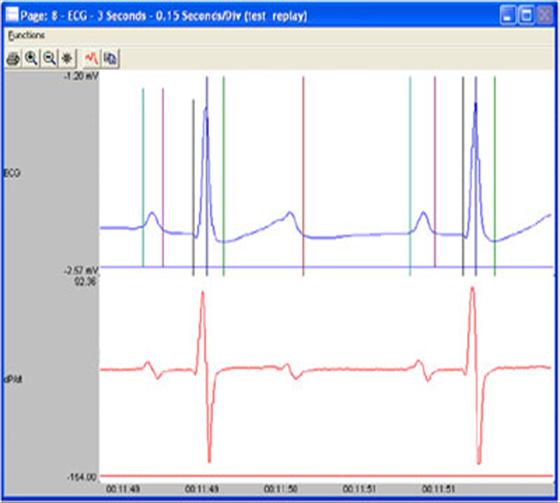
DSI has a long history of providing the essential tools to measure cardiovascular endpoints like blood pressure, heart rate, and ECG in both Safety Pharmacology and Toxicology studies. DSI also provides GLP-compliant systems for acquisition and reporting.
Small Animal Telemetry
PhysioTel HD-X and HD-S series telemetry implants are used in small animals such as mice, rats, ferrets or guinea pigs. These implants are capable of measuring pressure, temperature, activity and biopotential signals.

Large Animal Telemetry
PhysioTel Digital M and L series implants are designed for single use or long-term implantation in animals of rabbit size or larger. These implants are capable of measuring pressure, temperature, activity and biopotential signals.
 Ponemah Software
Ponemah Software
Ponemah Analysis Modules are trusted by researchers worldwide to discover new insights into their research applications. Ponemah integrates validated algorithms to reliably obtain accurate, consistent results and quickly deliver these results to you. Operate with full compliance within GLP environments with the Ponemah Data Security Option.
CNS Solutions
Small Animal CNS Solution
Experience the versatility of recording any combination of two biopotential channels (typically EEG and EMG), plus temperature and activity with the PhysioTel™ HD-X02 and HD-S02.

Hardwired Monitoring System
DSI’s hardwired (tethered) solutions provide a minimally invasive method to offer continuous measurement (EEG, EMG, EOG, etc.) during neuroscience studies with small animals. Tethered solutions allow monitoring of up to 12 EEG/EMG channels per animal.

Large Animal
Get the data you need by recording any combination of up to four biopotential channels (typically EEG, EMG, or ECG), plus temperature and activity with the PhysioTel™ Digital L03 and L04.
 NeuroScore Software
NeuroScore Software
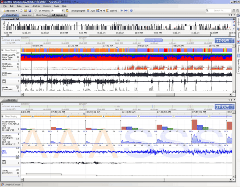
After data acquisition has taken place, DSI’s NeuroScore™ software can be used to efficiently analyze chronic data sets common to neuroscience studies. This modular platform offers sleep scoring, seizure detection, video synchronization, and batchprocessing capabilities.
FinePointe Respiratory System
FinePointe Software is powerful and easy-to-use for collecting, analyzing, and reporting life science data. Fully network-enabled, the smart design minimizes required user interaction while wizards walk users through necessary procedures.
Whole Body Plethysmography
When conscious, unrestrained approaches are desired, FinePointe whole body plethysmography allows for respiratory endpoints to be obtained. With the use of a bias flow supply, longer data collection periods can be obtained; allowing for evaluation of compounds that might have an effect beyond 4-6 hours post dose.
 Head-Out Plethysmography
Head-Out Plethysmography
Using Allay restraints, animals are comfortably and properly secured in plethysmographs without compromising the thorax to obtain consistent and reliable direct flow measurements.
 Resistance and Compliance
Resistance and Compliance
FinePointe resistance and compliance hardware helps researchers measure an animal’s airflow and lung pressure. This anesthetized approach allows for direct measurement pulmonary function as a follow up study to the core battery requirements.
 Non-Invasive Airway Mechanics Station
Non-Invasive Airway Mechanics Station
Conscious follow up studies for safety pharmacology and longitudinal toxicology studies alike can take advantage of the FinePointe non-invasive airway mechanics station. Using specially designed Allay restraint and double chamber plethysmography, nasal and thoracic flow can be monitored simultaneously to derived specific airway resistance.

Want to Learn More?
Click on one of the links below to receive more information about our current solutions.
Cardiovascular Solutions,
Glucose Metabolism,
Respiratory Solutions,
Neuroscience Solutions
Follow Us
On
LinkedIn,
Facebook and
Twitter to receive up to date information about Safety Pharmacology research using DSI solutions and product releases.