Chronic Pleural Pressure
In respiratory physiology, lung compliance and resistance to lung airflow are used as critical measures of lung function. Compliance is determined by measuring the transpulmonary pressure (i.e., pleural pressure minus mouth pressure) required to maximally inflate the lung. Resistance to airflow is usually determined by measuring the transpulmonary pressure required to generate maximal airflow during inspiration and/or expiration. To measure pleural pressure, pressure-sensitive probes have been inserted directly into the pleural cavity or advanced into the thoracic esophagus. Both techniques work well in acute, anesthetized preptions, but lung damage, tissue growth and encapsulation of the pressure-sensitive catheter have limited their use in chronic preptions. The group of Dr. Dennis J. Murphy at SmithKline Beecham Pharmaceuticals (King of Prussia, PA) described a novel surgical procedure, enabling long-term chronic measurements in conscious, freely moving rats. The researchers used adult, male Sprague Dawley rats (BW 340-609 g, 12-15 wks of age). Under isoflurane anesthesia, the abdominal cavity was opened by an ~ 5cm long midline incision. After retracting the lobes of the liver, the esophagus was isolated ~ 2cm from its junction with the diaphragm. A small incision was then made through the serosal (outer) layer of the esophagus, through which a 22-gauge needle was carefully inserted between the serosal and muscularis layers. This needle was tunneled cranially past the junction with the diaphragm and into the thoracic cavity. After retracting the needle, the catheter of a model TA11PA-C40 pressure transmitter was advanced through the channel prepared by the needle, until maximal pleural pressure changes were monitored. The catheter was then fixed in place at the entry point with tissue adhesive. The body of the transmitter was secured to the abdominal wall during surgical closure of the abdominal musculature.
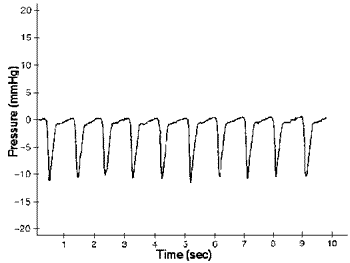
A typical tracing of pleural pressure changes in a conscious, non-restrained rat. Recorded with the subpleural catheter of a TA11PA-C40 pressure transmitter. Sample frequency: 500Hz.
After recovery, end-expiratory pressures of 0 +/- 5 mmHg, and end-inspiratory pressures of -5 to -15 mmHg were measured (Fig. 1), values that have previously been described in spontaneously breathing rats, guinea pigs and hamsters. No evidence of damping or loss of the pressure signal was seen in 4 of the 5 implanted rats during 14 weeks of repeated measurements. In one rat the amplitude of the pressure changes remained constant for 3 weeks, but was reduced by >50% through weeks 4-14. At necropsy, the catheters of the 4 rats with undamped pressures were found intact beneath the serosal surface, while the catheter in the 5th rat had penetrated the serosal and pleural surfaces and was encapsulated. Comparison of pleural pressure changes obtained simultaneously with the transmitter and with an intrapleurally implanted catheter in anesthetized, spontaneously breathing rats demonstrated the accuracy of the new technique. Linear regression analysis showed a close correlation between the pressures measured by the two methods (see Fig. 2).
Pressure changes in individual breaths of an anesthetized rat Measured simultaneously subpleurally by telemetry (X-axis) and intrapleurally by an acute implanted pressure catheter (Y-axis). Solid line, regression line; dashed lines, 95% confidence limits of the regression line. (Used with permission)
Whereas these results look very promising for chronically monitoring changes in lung compliance and airflow resistance in conscious, freely moving rats, the authors warn that the application of this novel technique in other species has to be investigated further, in particular with respect to the effect of esophageal wall thickness and damping of the pressure signal.
A novel method for chronic measurement of pleural pressure in conscious rats. Murphy D.J. Renninger J.P. Gossett K.A. J. Pharmacol. Toxicol. Methods 1998 39 137-141